Gen-Orph
Mettre à disposition des patients et des médecins, des médicaments pour les maladies et d'autres pathologies
Notre mission
L’accès facilité à des médicaments utiles et abordables.

Gen.Orph concentre ses efforts sur la mise à disposition des patients et du corps médical international de médicaments orphelins utiles et abordables en vue du traitement de certaines maladies rares, de médicaments classiques, conservant un potentiel médical et économique attractif ainsi que des médicaments innovants pour des maladies fréquentes.
Produits
Produits orphelins pour maladies rares.

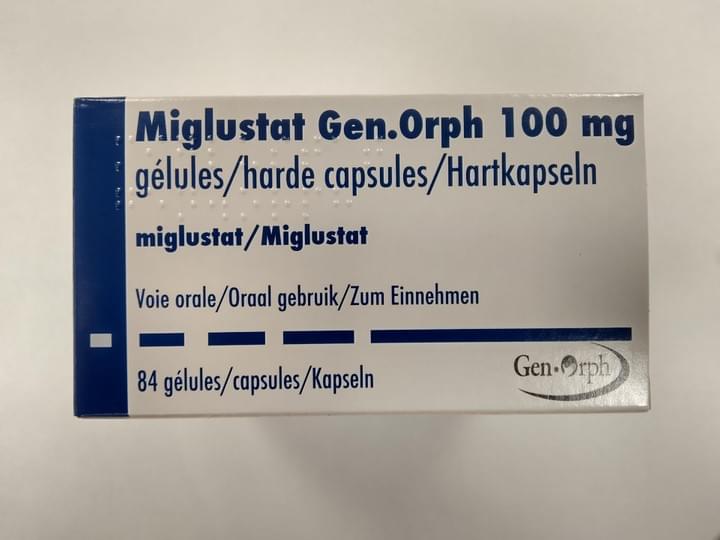
Miglustat GEN-ORPH
Traitement de maladies métaboliques héréditaires par déficit enzymatique congénital.
Indications :
- Traitement par voie orale des patients adultes atteints de la maladie de Gaucher de type 1 légère à modérée. Ne doit être utilisé que pour le traitement des patients chez lesquels la thérapie de remplacement enzymatique ne convient pas (voir rubrique 4.4 et 5.1 du RCP).
- Traitement des manifestations neurologiques progressives des patients adultes et des enfants atteints de maladie Niemann-Pick Type C (voir rubrique 4.4 et 5.1 du RCP)
Enregistrement Européen centralisé
Miglustat Gen-Orph est commercialisé dans les pays suivants : France, Suède, Norvège, Danemark, Italie, Bulgarie, Allemagne, Portugal, Espagne, Royaume-Uni, Belgique, Luxembourg, Hongrie, Arabie Saoudite, Oman.
Prochainement : Mexique, Équateur, Lybie, République Tchèque

Ibuprofène GEN-ORPH
5 mg / ml - Solution injectable
Indications :
Traitement du canal artériel persistant hémodynamiquement significatif chez le nouveau-né prématuré d'âge gestationnel inférieur à 34 semaines
Enregistrement Européen centralisé - Mars 2024
Lancement prévu en 2024
Autres produits commercialisés par Gen.Orph

Agyrax
Dichlohydrate de meclozine
Indications :
Traitement symptomatique de la crise vertigineuse.
Prévention et traitement du mal des transports
Commercialisé en France, Belgique, Luxembourg, Nouvelle Zélande, Sénégal et Côte d'Ivoire

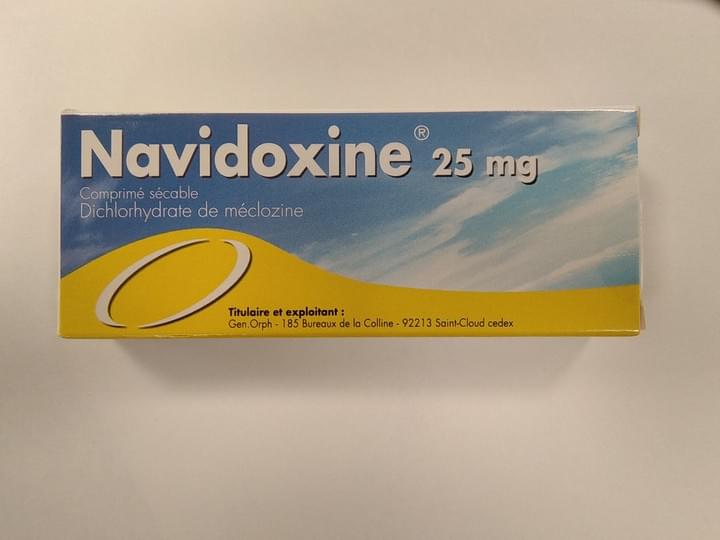
Navidoxine
Dichlohydrate de meclozine
Indication :
Prévention et traitement du mal des transports
Commercialisé :
France et Malte
Afin de consulter le résumé des caractéristiques produits, la notice, l'avis de la commission de transparence, se référer au lien suivant :
L'engagement qualité
Tous les médicaments peuvent avoir des effets indésirables. Si vous ressentez un quelconque effet indésirable, qu’il figure ou non dans la notice du produit, parlez-en à votre médecin ou à votre pharmacien. Ceci s’applique aussi à tout effet qui ne serait pas mentionné dans la notice du produit. Vous pouvez également déclarer les effets indésirables suspectés directement via le système national de déclaration : Agence Nationale de Sécurité du Médicament et des produits de santé (ANSM) et reséau des centres régionaux de pharmacovigilance de https://signalement.social-sante.gouv.fr
Notre laboratoire est à votre service pour vous délivrer toute information concernant nos spécialités.
Pour signaler un effet indésirable ou avoir une information médicale, vous pouvez nous joindre par courriel : PR@gen-orph.com ou par téléphone : +33(0)1.47.71.04.50
RGPD
GEN-ORPH respecte votre vie privée et protège vos données personnelles. Vos données personnelles seront traitées conformément à la notice d'information sur les données dans l'espace Economique Européen concernant l'information médicale, les réclamations Produit et la sécurité des médicaments. Pour toute question relative à l'utilisation par GEN-ORPH de vos données personnelles ou pour recevoir par e-mail ou courrier, veuillez-bien nous contacter par e-mail ou par téléphone : contact@gen-orph.com ou par téléphone : +33(0)1.47.71.04.50
Recherche
D'autres produits sont en cours de développement.

L'équipe
Etienne Jacob
Président Directeur Général
Fondateur
Docteur en Médecine
Pitié-Salpetriere, Paris
Harvard Business School – AMP
Patrice Layrac
Directeur Commercial
Associé
Fondateur
Doctorat en Gestion et Economie de l’université de Paris Dauphine et IEP INSEAD.
Eric Lille
Directeur Administratif
et Financier
Dr. A.M. Charrier
Directeur
Affaires Réglementaires
Etienne Horreard
Pharmacien Responsable
Florence Jomard
Pharmacien Responsable Intérimaire
Franck Grodenic
Directeur
Affaires Industrielles
Contactez nous
Genorph © 2017